Fletcher W Halliday1†, Tuomas Aivelo†, Pablo Beldomenico†, Aaron Blackwell†, Vanessa Ezenwa†, Anna Jolles†, Erin Gorsich†, Claudia Fichtel†, Charlotte Defolie†, Anni M Hämäläinen†, Josue H Rakotoniaina†, Eva Kaesler†, Corey Freeman-Gallant†, Michael Huffman†, Natalie Olifers†, Arnaldo M Júnior†, Elisa P de Araujo†, Rita de Cassia Bianchi†, Ana PN Gomes†, Paulo HD Cançado†, Matthew E Gompper†, & Paulo S D’Andrea†, Miroslava Soldanova†, Christopher Taylor†, Jason R Rohr
† contributed dataset
1Department of Evolutionary Biology and Environmental Studies, University of Zurich, Zurich, CH
Download the pdf of this document here.
A current frontier in disease ecology is understanding how interactions among parasite species influence their epidemics. Interactions among parasites can result when prior infection by one parasite alters host susceptibility to a second parasite, generating priority effects among parasite species within host individuals. The increasing number of laboratory studies that aim to measure priority effects highlights growing interest among disease ecologists to understand these processes. Yet, laboratory studies, which are implemented at the scale of host individuals, are poorly suited to understand parasite epidemics, which occur at the scale of host populations, posing a key challenge for disease ecologists. One way to overcome this challenge is to mark and then repeatedly recapture sentinel hosts in the field, then analyze the resulting data using longitudinal regression models (Fenton et al. 2014; Hellard et al. 2015; Halliday et al. 2017, 2018). The purpose of this project was to implement this approach across empirical systems to test two broad questions:
- Is disease risk influenced by infection sequence during natural epidemics (i.e., do parasites generally experience/exhibit priority effects)?
- To what extent do these within-host priority effects depend on traits of individual parasite species?
To evaluate the role of within-host priority effects during parasite epidemics, we compiled longitudinal datasets of coinfection in host individuals across 13 host and 110 parasite species, including over 25,000 observations of host plants, primates, ungulates, small mammals, birds, and invertebrates. To evaluate the role of within-host priority effects, we performed time-until event analysis, specifically testing whether infection sequence among co-occurring parasites influenced their risk of infection (e.g., Halliday et al. 2017, 2019). Surprisingly, the sequence of infection predicted fewer than 10% of the more than 1000 pairwise combinations of potentially interacting parasites. This rarity of within-host priority effects may result from a lack of natural variation in infection sequence, indicating that within-host priority effects may be a less common driver of parasite epidemics than previously thought, and highlighting the need for experimental manipulations of parasite infection in the field (e.g., Ezenwa & Jolles 2015; Pedersen & Fenton 2015; Halliday et al. 2017). Showing weak support for ecological theory, parasites with a high degree of niche overlap were slightly more sensitive to priority effects than parasites with a low degree of niche overlap, and this effect was stronger for parasites with narrow niche requirements than with wide niche-requirements. However, in contrast with theory, the frequency of priority effects could not be explained by a parasite’s impact on its host. Together, these results indicate that priority effects may be less commonly detected during natural epidemics than previously expected, and that within-host priority effects among parasites may not necessarily follow the same ecological rules of community assembly as their free-living counterparts.
The database:
We compiled a database of longitudinal surveys of host individuals (i.e., mark-recapture data), where hosts were unmanipulated (sentinels were ok), and surveyed for infection by multiple parasites. This study focused on interspecific interactions among parasites rather than intraspecific interactions among strains of the same parasite species. The final database includes plants, invertebrate, and vertebrate hosts (Table 1).
| Table 1. Contributing authors and associated host datasets | ||
| Contributors | Host species | Host taxon |
| T. Aivelo | Microcebus rufus | Primate |
| P. Beldomenico | Hydrochoerus hydrochaeris | Rodent |
| A. Blackwell | Homo sapiens | Human |
| V. Ezenwa, A. Jolles, E. Gorsich | Syncerus caffer | Wild ungulate |
| AM Hämäläinen, JH Rakotoniaina, E. Kaesler, C. Kraus, P. Kappeler | Microcebus murinus | Primate |
| C. Fichtel, C. Defolie | Eulemur rufifrons | Primate |
| C. Freeman-Gallant | Geothlypis trichas | Bird |
| F. Halliday | Festuca arundinacea | Plant |
| M. Huffman | Pan troglodytes | Primate |
| N. Olifers, AM Júnior, EP de Araujo , R de Cassia Bianchi, APN Gomes, PHD Cançado, ME Gompper, & PS D’Andrea | Nasua nasua & Cerdocyon thous | Carnivore |
| M. Soldanova | Lymnaea stagnalis | Invertebrate |
| C. Taylor | Microtus agrestis | Rodent |
A standard measurement of within-host priority effects across systems:
To facilitate comparisons across systems, data were analyzed using a common analytical method (described in Halliday et al 2017). Briefly, to detect priority effects, we recorded the sequence of infection by each parasite on each host individual. Time in days since the first survey of each host individual was used as a proxy for exposure to parasite propagules. To model within-host priority effects, we constructed a series of models following Halliday et al (2017). These models explicitly measure priority effects by testing whether the sequence of infection on an individual host influences the rate of infection by each parasite. Each model included one dependent variable pertaining to one parasite species (“the focal parasite”). To explicitly model priority effects, we used Cox Regression with Firth’s Penalized Likelihood to reduce bias from factors exhibiting (quasi)complete separation (Heinze & Schemper 2002) from the R package, coxphf (Ploner & Heinze 2015), to estimate the probability of a host transitioning from uninfected to infected. Specifically, the dependent variable in each model was time to infection, modeled as the transition rate from uninfected to infected as a function of exposure time. We modeled time to infection resulting from a baseline rate of infection shared by all individuals and modified by the infection status of the host by each other parasite during the previous survey (treated as a time-varying coefficient). Exponentiated coefficients are interpreted as multiplicative changes in infection rate, providing a standardized estimate of priority effects across study systems. Statistically significant (p<0.05) positive and negative coefficients were interpreted as evidence of facilitative and antagonistic priority effects, respectively, whereas non-significant coefficients were interpreted as lacking sufficient evidence of a priority effect occurring.
Our hypotheses:
Hypothesis 1 – Niche overlap. Priority effects are expected to occur more commonly among species with a high degree of niche overlap (Vannette & Fukami 2014). A host comprises the entire niche available to parasites during infection (Kuris et al. 1980; Rynkiewicz et al. 2015), and thus coinfecting parasites often exhibit high niche overlap (Sousa & Zoologist 1992; Graham 2008; Seabloom et al. 2015). The degree of niche overlap among parasites may additionally vary depending on whether parasites share common vectors, employ similar feeding strategies, or share infection sites. We hypothesized that such parasites would therefore more commonly experience priority effects.
Hypothesis 2 – Parasite virulence. Priority effects are expected to occur when early arriving species have a high impact on shared resources with later arriving species (Vannette & Fukami 2014). Parasites require host resources for survival, growth, and reproduction (Lafferty & Kuris 2002). We hypothesized that early-arriving parasites that were more damaging to their hosts would more commonly experience priority effects.
Hypothesis 3 – Host breadth. Priority effects are expected occur when the late arriving species have high requirements for shared resources with early arriving species (Vannette & Fukami 2014). Late arriving species with narrow niche requirements may therefore experience the strongest priority effects. For free-living species, the breadth of niche requirements may manifest as generality in a species’ use of habitat types or tolerance to environmental conditions. For parasites, the breadth of niche requirements may be related to host specificity, defined as the ability to infect multiple unrelated host taxa (Barrett et al. 2009; Park et al. 2018). We hypothesized that late-arriving host specialists would therefore more commonly experience priority effects.
Analytical methods
Data analysis was performed using R version 3.5.2 (R Core Team 2015). To test whether parasites that shared similar traits more commonly experienced priority effects (Hypothesis 1), we used a series of generalized linear mixed models using the lme4 package, version 1.1-20 (Bates et al. 2014), treating the source dataset and host species (Table 1) as random intercepts in each model. The dependent variable in each model was whether or not a pairwise combination of parasites exhibited a priority effect (0,1), and the independent variables where whether or not parasites were the same type (e.g., both macroparasites), shared a common infection site (e.g., both gastrointestinal), or shared the same transmission mode (e.g., both tick-vectored). Data for every trait was not available for every parasite species (Table S1), so in order to maximize the number of studies contributing to the analysis, each independent variable was therefore tested in a separate analysis. We used inverse-variance weighting based on the number of surveys per host to give more explanatory weight to studies with a greater number of surveys of each host individual. However, because this weighting strongly influenced the results of the analyses, we report results from both weighted and unweighted analyses.
To test whether early-arriving parasites that were more damaging to their hosts would more commonly experience priority effects, we again tested whether or not a pairwise combination of parasites exhibited a priority effect, this time as a function of host virulence. We coded host virulence as 1 if the parasite had few reported adverse effects on the host, 2 if the parasite was known to moderately reduce host fitness, and 3 if the parasite was known to seriously impact host survival (e.g., infections frequently leading to host mortality).
To test whether late-arriving host specialists more commonly experienced priority effects, we used a similar, weighted generalized linear mixed model in lme4, testing whether or not a pairwise combination of parasites exhibited a priority effect as a function of the host specificity of the late-arriving parasite (generalist = known to infect more than one host species; specialist = known to infect only a single host species).
We finally tested whether niche-overlap lead to stronger priority effects among late-arriving host specialists by analyzing the interaction coefficient for each pairwise combination of parasites as a dependent variable. Because more closely related species tend to exhibit higher niche-overlap, we used the phylogenetic relatedness of parasites as a metric of niche-overlap, and modeled its interaction with host specificity of the late-arriving parasite and their main effects as independent variables in this analysis. Parasites were defined as “closely related” if they shared at least the same order, and “distantly related” if they were from different orders or more distantly related.
Results
Contrary to expectations, we found surprisingly limited evidence of priority effects across our database. Specifically, out of 1128 potential pairwise interactions, we detected only 98 interactions resulting from prior infection (i.e., priority effects). 64 were facilitative (prior infection by one parasite species facilitated subsequent infection by another species), 34 interactions were antagonistic (prior infection by one parasite species inhibited infection by a subsequent species), and the remaining 1030 lacked sufficient evidence to support a priority effect occurring. This surprising result may stem from a lack of variation in infection sequence among study systems. Early infections were often dominated by one or a few parasite species (Fig. 1), potentially resulting from variation in parasite phenology (Halliday et al. 2017) or variation in parasite transmission rates (Clay et al. 2019). Together, these results point to the need for studies that systematically manipulate infection sequence during natural epidemics, potentially through the use of pesticides or anti-parasite treatments of hosts (Pedersen & Fenton 2015).

We found weak support for the hypothesis that parasites with high niche overlap would more commonly experience priority effects. Specifically, when weighted by the number of samples per host individual, parasites of the same type (e.g., both macroparasites), parasites that infected the same site (e.g., gastrointestinal parasites), and parasites with the same transmission mode (e.g., both tick vectored), all experienced significantly more priority effects than parasites of different types, different sites, and different transmission modes (p= 0.006, p<0.001, and p = 0.004, respectively; Fig 2). However, these effects were of small magnitude and were not statistically significant when the same analyses were performed with unweighted data (p = 0.98, p = 0.48, p = 0.68, respectively). We found no support for the hypothesis that early-arriving parasites that were more damaging to their hosts would more commonly experience priority effects. Even though the weighted regression indicated that parasite virulence significantly predicted the priority effects (p < 0.001), the direction of this effect was not consistent with our prediction, with moderately virulent parasites exhibiting the lowest rate of priority effects (Fig 2). The effect of host virulence on the probability of observing a priority effect was not significant when the analysis was performed with unweighted data (p = 0.25).
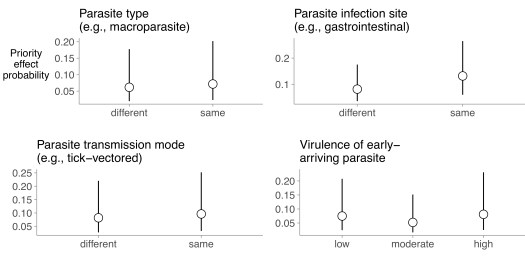
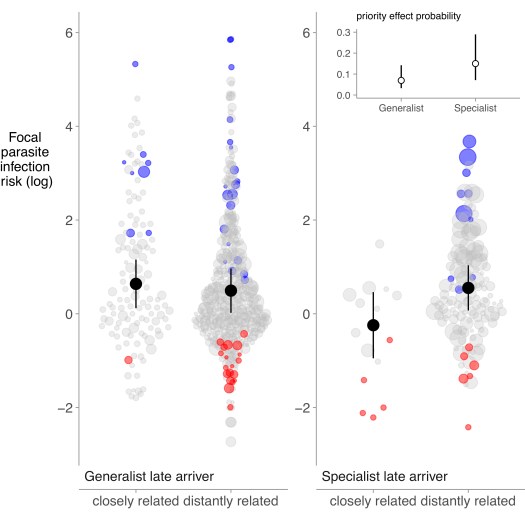
We found strong evidence that later arriving parasites with narrow host breadth (e.g., specialists) more commonly experienced priority effects than parasites with wider host breadth (e.g., generalists; weigthed p < 0.001, unweighted p = 0.007; Fig. 3 – inset). Furthermore, among host specialists, more closely related parasites tended to exhibit more antagonistic interactions (p = 0.003). Together, these results indicate that priority effects among parasites may be less common than expected based on laboratory studies, and that priority effects are rarely consistent with predictions from ecological theory.
References
Barrett, L.G., Kniskern, J.M., Bodenhausen, N., Zhang, W. & Bergelson, J. (2009). Continua of specificity and virulence in plant host–pathogen interactions: causes and consequences. New Phytol., 183, 513–529.
Bates, D., Mächler, M., Bolker, B. & Walker, S. (2014). Fitting Linear Mixed-Effects Models using lme4. J. Stat. Softw., 67, 1–48.
Clay, P.A., Cortez, M.H., Duffy, M.A. & Rudolf, V.H.W. (2019). Priority effects within coinfected hosts can drive unexpected population‐scale patterns of parasite prevalence. Oikos, 128, 571–583.
Ezenwa, V. & Jolles, A. (2015). Opposite effects of anthelmintic treatment on microbial infection at individual versus population scales. Science (80-. ).
Fenton, A., Knowles, S.C.L., Petchey, O.L. & Pedersen, A.B. (2014). The reliability of observational approaches for detecting interspecific parasite interactions: comparison with experimental results. Int. J. Parasitol., 44, 437–45.
Graham, A.L. (2008). Ecological rules governing helminth-microparasite coinfection. Proc. Natl. Acad. Sci. U. S. A., 105, 566–70.
Halliday, F.W., Heckman, R.W., Wilfahrt, P.A. & Mitchell, C.E. (2019). Past is prologue: host community assembly and the risk of infectious disease over time. Ecol. Lett., 22, 138–148.
Halliday, F.W., Umbanhowar, J. & Mitchell, C.E. (2017). Interactions among symbionts operate across scales to influence parasite epidemics. Ecol. Lett., 20, 1285–1294.
Halliday, F.W., Umbanhowar, J. & Mitchell, C.E. (2018). A host immune hormone modifies parasite species interactions and epidemics: insights from a field manipulation. Proc. R. Soc. B Biol. Sci., 285, 20182075.
Heinze, G. & Schemper, M. (2002). A solution to the problem of separation in logistic regression. Stat. Med., 21, 2409–2419.
Hellard, E., Fouchet, D., Vavre, F. & Pontier, D. (2015). Parasite-Parasite Interactions in the Wild: How To Detect Them? Trends Parasitol.
Kuris, A.M., Blaustein, A.R. & Alio, J.J. (1980). Hosts as Islands. Am. Nat., 116, 570–586.
Lafferty, K.D. & Kuris, A.M. (2002). Trophic strategies, animal diversity and body size. Tree, 17, 507–513.
Park, A.W., Farrell, M.J., Schmidt, J.P., Huang, S., Dallas, T.A., Pappalardo, P., et al. (2018). Characterizing the phylogenetic specialism-generalism spectrum of mammal parasites. Proc. R. Soc. B Biol. Sci., 285, 20172613.
Pedersen, A.B. & Fenton, A. (2015). The role of antiparasite treatment experiments in assessing the impact of parasites on wildlife. Trends Parasitol., 31, 200–11.
Ploner, M. & Heinze, G. (2015). coxphf: Cox regression with Firth’s penalized likelihood. R Found. Stat. Comput.
R Core Team. (2015). R: a language and environment for statistical computing | GBIF.ORG.
Rynkiewicz, E.C., Pedersen, A.B. & Fenton, A. (2015). An ecosystem approach to understanding and managing within-host parasite community dynamics. Trends Parasitol., 31, 212–221.
Seabloom, E.W., Borer, E.T., Gross, K., Kendig, A.E., Lacroix, C., Mitchell, C.E., et al. (2015). The community ecology of pathogens: Coinfection, coexistence and community composition. Ecol. Lett., 18, 401–415.
Sousa, W.P. & Zoologist, A. (1992). Interspecific Interactions among Larval Trematode Parasites of Freshwater and Marine Snails Interspecific Interactions Among Larval Trematode Parasites of Freshwater and Marine Snails1. Interactions, 32, 583–592.
Vannette, R.L. & Fukami, T. (2014). Historical contingency in species interactions: Towards niche-based predictions. Ecol. Lett., 17, 115–124.
Table S1. Parasites and their associated traits
| Parasite | Type | Virulence | Specialist | Route | Mode | Helminth | Intracellular |
| Anaplasma centrale | microparasite | Low | Generalist | Complex | Tick vectored | Not Helminth | Intracellular |
| Ascaris lumbricoides | macroparasite | Low | Specialist | Direct | Ingestion | Helminth | Not intracellular |
| Anaplasma marginale | microparasite | Medium | Generalist | Complex | Tick vectored | Not Helminth | Intracellular |
| Anaplasma phagocytophilum | microparasite | Medium | Generalist | Complex | Tick vectored | Not Helminth | Intracellular |
| Acanthocephala | macroparasite | Low | Complex | Ingestion | Helminth | Not intracellular | |
| Amblyomma cajennense | ectoparasite | Low | Generalist | Direct | Free-living | Not Helminth | Not intracellular |
| Amblyomma ovale | ectoparasite | Low | Generalist | Direct | Free-living | Not Helminth | Not intracellular |
| Amblyomma parvum | ectoparasite | Low | Generalist | Direct | Free-living | Not Helminth | Not intracellular |
| Apicomplexa | microparasite | Low | Complex | Not Helminth | Intracellular | ||
| Ascaridae | macroparasite | Low | Direct | Ingestion | Helminth | Not intracellular | |
| Ascaridida | macroparasite | Low | Direct | Ingestion | Helminth | Not intracellular | |
| Aspidoderidae | macroparasite | Low | Direct | Ingestion | Helminth | Not intracellular | |
| Australapatemon burti | macroparasite | Medium | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Australapatemon minor | macroparasite | Medium | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Babesia | microparasite | Low | Generalist | Complex | Tick vectored | Not Helminth | Intracellular |
| Ballantidium | microparasite | Low | Direct | Ingestion | Not Helminth | Not intracellular | |
| Bartonella | microparasite | Medium | Generalist | Complex | Flea vectored | Not Helminth | Intracellular |
| Brucella abortus | microparasite | Medium | Generalist | Direct | Ingestion | Not Helminth | Intracellular |
| Callistoura | macroparasite | Low | Direct | Ingestion | Helminth | Not intracellular | |
| Capillaria | macroparasite | Low | Complex | Ingestion | Helminth | Not intracellular | |
| Cestoda | macroparasite | Low | Complex | Ingestion | Helminth | Not intracellular | |
| Conoidasida | microparasite | Low | Direct | Ingestion | Not Helminth | Intracellular | |
| Conoidasida | microparasite | Low | Direct | Ingestion | Not Helminth | Intracellular | |
| Colletotrichum cereale | microparasite | Low | Generalist | Direct | Rain splash | Not Helminth | Not intracellular |
| Cooperia | macroparasite | Low | Generalist | Direct | Ingestion | Helminth | Not intracellular |
| Cotylurus cornutus | macroparasite | Medium | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Cowpox virus | microparasite | Medium | Generalist | Direct | Direct contact | Not Helminth | Intracellular |
| Dicrocoeliidae | macroparasite | Low | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Diphyllobothriidae | macroparasite | Low | Complex | Ingestion | Helminth | Not intracellular | |
| Diplostomum pseudospathaceum | macroparasite | Low | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Dipylidiidae | macroparasite | Low | Complex | Ingestion | Helminth | Not intracellular | |
| Ehrlichia ruminantium | microparasite | Medium | Generalist | Complex | Tick vectored | Not Helminth | Intracellular |
| Anaplasma | microparasite | Low | Specialist | Complex | Tick vectored | Not Helminth | Intracellular |
| Echinocleus hydrochaeri | macroparasite | Low | Specialist | Complex | Ingestion | Helminth | Not intracellular |
| Echinoparyphium aconiatum | macroparasite | Medium | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Echinoparyphium recurvatum | macroparasite | Medium | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Echinostoma revolutum | macroparasite | Medium | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Eimeria boliviensis | microparasite | Medium | Specialist | Direct | Ingestion | Not Helminth | Intracellular |
| Eimeria hydrochaeri | microparasite | Medium | Specialist | Direct | Ingestion | Not Helminth | Intracellular |
| Eimeria | microparasite | Medium | Specialist | Direct | Ingestion | Not Helminth | Intracellular |
| Entamoeba | microparasite | Medium | Direct | Ingestion | Not Helminth | Not intracellular | |
| Fasciolidae | macroparasite | Low | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Siphonaptera | ectoparasite | Low | Generalist | Direct | Free-living | Not Helminth | Not intracellular |
| Giardia lamblia | microparasite | Medium | Generalist | Direct | Ingestion | Not Helminth | Not intracellular |
| Haemonchus | macroparasite | Medium | Direct | Ingestion | Helminth | Not intracellular | |
| Ancylostomatidae | macroparasite | Low | Specialist | Direct | Free-living | Helminth | Not intracellular |
| Hymenolepis | macroparasite | Low | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Hypoderaeum conoideum | macroparasite | Low | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Laelapidae | ectoparasite | Low | Direct | Free-living | Not Helminth | Not intracellular | |
| Lemuricola | macroparasite | Low | Specialist | Direct | Ingestion | Helminth | Not intracellular |
| Lemuricola | macroparasite | Low | Specialist | Direct | Ingestion | Helminth | Not intracellular |
| Lemurostrongylus | macroparasite | Low | Specialist | Direct | Ingestion | Helminth | Not intracellular |
| Leucocytozoon | microparasite | Low | Generalist | Complex | Blackfly vectored | Not Helminth | Intracellular |
| Listrophorus | ectoparasite | Low | Generalist | Direct | Free-living | Not Helminth | Not intracellular |
| Phthiraptera | ectoparasite | Low | Generalist | Direct | Free-living | Not Helminth | Not intracellular |
| Metagonymus | macroparasite | Low | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Hystrichopsyllidae | ectoparasite | Low | Generalist | Direct | Free-living | Not Helminth | Not intracellular |
| Moliniella anceps | macroparasite | Low | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Myobiidae | ectoparasite | Low | Generalist | Direct | Free-living | Not Helminth | Not intracellular |
| Neotrichodectes pallidus | ectoparasite | Low | Specialist | Direct | Free-living | Not Helminth | Not intracellular |
| Notocotylus attenuatus | macroparasite | Low | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Oesophagostomum | macroparasite | Medium | Generalist | Direct | Ingestion | Helminth | Not intracellular |
| Opisthioglyphe ranae | macroparasite | Low | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Opisthorchis | macroparasite | Low | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Oesophagostomum stephanostomum | macroparasite | Medium | Generalist | Direct | Ingestion | Helminth | Not intracellular |
| Oxyuridae | macroparasite | Low | Direct | Ingestion | Helminth | Not intracellular | |
| Oxyuridae | macroparasite | Low | Direct | Ingestion | Helminth | Not intracellular | |
| Oxyuridae | macroparasite | Low | Direct | Ingestion | Helminth | Not intracellular | |
| Pararhabdonema | macroparasite | Low | Generalist | Direct | Ingestion | Helminth | Not intracellular |
| Paryphostomum radiatum | macroparasite | Low | Generalist | Complex | Ingestion | Helminth | Not intracellular |
| Physalopteridae | macroparasite | Low | Complex | Ingestion | Helminth | Not intracellular | |
| Plagiorchis elegans | macroparasite | Low | Specialist | Complex | Ingestion | Helminth | Not intracellular |
| Plasmodium | microparasite | Medium | Specialist | Complex | Mosquito vectored | Not Helminth | Intracellular |
| Protozoophaga obesa | macroparasite | Low | Specialist | Direct | Ingestion | Helminth | Not intracellular |
| Strongyloides | macroparasite | Low | Generalist | Direct | Free-living | Helminth | Not intracellular |
| Caenorhabditis | macroparasite | Low | Direct | Ingestion | Helminth | Not intracellular | |
| Strongyloidae | macroparasite | Low | Generalist | Direct | Ingestion | Helminth | Not intracellular |
| Chromadorea | macroparasite | Low | Helminth | Not intracellular | |||
| Enterobius | macroparasite | Low | Direct | Ingestion | Helminth | Not intracellular | |
| Panagrellus | macroparasite | Low | Direct | Ingestion | Helminth | Not intracellular | |
| Puccinia coronata | microparasite | Low | Specialist | Complex | Airborne | Not Helminth | Not intracellular |
| Rhizoctonia solani | macroparasite | Medium | Generalist | Direct | Rain splash | Not Helminth | Not intracellular |
| Schistosoma | macroparasite | Medium | Generalist | Complex | Free-living | Helminth | Not intracellular |
| Schistosomatidae | macroparasite | Medium | Generalist | Complex | Free-living | Helminth | Not intracellular |
| Strongyloides fuelleborni | macroparasite | Low | Generalist | Direct | Ingestion | Helminth | Not intracellular |
| Strongiloides | macroparasite | Low | Generalist | Complex | Free-living | Helminth | Not intracellular |
| Strongyloidae | macroparasite | Low | Generalist | Complex | Free-living | Helminth | Not intracellular |
| Strongyloidae | macroparasite | Low | Generalist | Complex | Free-living | Helminth | Not intracellular |
| Strongyloidae | macroparasite | Low | Generalist | Complex | Free-living | Helminth | Not intracellular |
| Strongiloides chapini | macroparasite | Low | Generalist | Complex | Free-living | Helminth | Not intracellular |
| Subulura | macroparasite | Low | Complex | Ingestion | Helminth | Not intracellular | |
| Subulura | macroparasite | Low | Complex | Ingestion | Helminth | Not intracellular | |
| Theileria mutans | microparasite | Low | Generalist | Complex | Tick vectored | Not Helminth | Intracellular |
| Theileria parva | microparasite | High | Generalist | Complex | Tick vectored | Not Helminth | Intracellular |
| Theileria sable | microparasite | Low | Generalist | Complex | Tick vectored | Not Helminth | Intracellular |
| Theileria sp (buffalo) | microparasite | Medium | Generalist | Complex | Tick vectored | Not Helminth | Intracellular |
| Trichuris trichiura | macroparasite | Low | Specialist | Direct | Ingestion | Helminth | Not intracellular |
| Theileria velifera | microparasite | Low | Generalist | Complex | Tick vectored | Not Helminth | Intracellular |
| Mycobacterium tuberculosis | microparasite | Medium | Specialist | Direct | Airborne | Not Helminth | Intracellular |
| Thysanotaenia | macroparasite | Low | Specialist | Complex | Ingestion | Helminth | Not intracellular |
| Ixodida | ectoparasite | Low | Generalist | Direct | Free-living | Not Helminth | Not intracellular |
| Trematoda | macroparasite | Low | Complex | Helminth | Not intracellular | ||
| Trichinellidae | macroparasite | Low | Complex | Ingestion | Helminth | Not intracellular | |
| Trichobilharzia szidati | macroparasite | Low | Generalist | Complex | Free-living | Helminth | Not intracellular |
| Trichodectes canis | ectoparasite | Low | Generalist | Direct | Free-living | Not Helminth | Not intracellular |
| Trichostrongyloidea | macroparasite | Low | Direct | Ingestion | Helminth | Not intracellular | |
| Trichuris | macroparasite | Low | Specialist | Direct | Ingestion | Helminth | Not intracellular |
| Trichuris cutillasae | macroparasite | Low | Specialist | Direct | Ingestion | Helminth | Not intracellular |
| Trichuris | macroparasite | Low | Specialist | Direct | Ingestion | Helminth | Not intracellular |
| Trypanosoma | microparasite | Medium | Generalist | Complex | Tsetse fly vectored | Not Helminth | Intracellular |
| Trichuris trichiura | macroparasite | Low | Specialist | Direct | Ingestion | Helminth | Not intracellular |







