Our world is undergoing rapid shifts that touch our everyday lives, including the emergence and spread of infectious diseases, changing species diversity, and a changing climate. Understanding how these processes interact with one another will be the key to predicting and ameliorating their impacts on food production systems, wildlife, and humans. In the context of infectious disease, limiting the impacts of global change requires the ability to predict when and where crops, livestock, wildlife, and humans are at the greatest risk of becoming infected. Disease ecology provides a framework for achieving this goal through careful examination of interactions among hosts, parasites, and the environment. I apply this approach across scales, from interactions among parasites within individual host organs all the way up to global patterns of biodiversity and disease risk, using experiments, large-scale field surveys, and data syntheses.

Within-host drivers of infectious disease
Interactions among parasites are determined by environmental factors like temperature that can drive parasite phenology, and by characteristics of hosts, like the host immune response to infection. Using model hosts like tall fescue (Lolium arundinaceum) and ribwort plantain (Plantago lanceolata), I try to understand how these interactions, which occur within host individuals, alter parasite epidemics, which occur across host populations.

Host community-level drivers of infectious disease
The risk of an individual becoming infected can be determined by characteristics of host species, like host provenance, relatedness, or growth rate. Disease risk can also be determined by environmental factors like resource supply and temperature that directly regulate parasite growth and reproduction. The environment can also indirectly affect disease via ecological filtering by determining the trajectory of community assembly, and thereby altering which host species are most abundant in a given time and place. Using experimental manipulations of herbaceous plant communities, I try to unravel the relative contributions of these processes to disease risk in host communities.
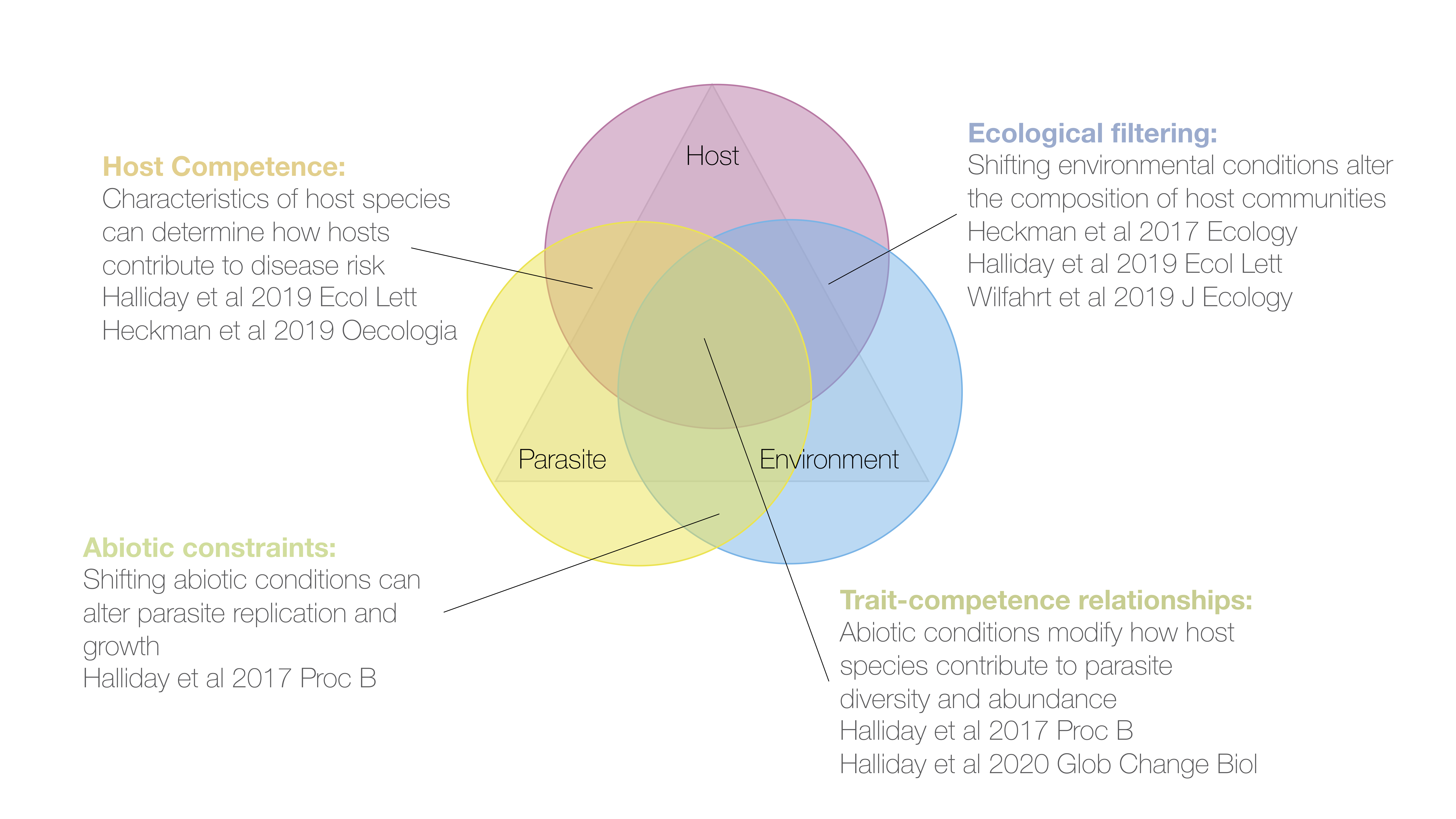

I am currently exploring these questions along a 1100m elevational gradient in southeastern Switzerland. By integrating manipulative experiments with field surveys along this gradient, I hope to provide an unprecedented synthesis of how host and parasite communities respond to global change.
Data synthesis
Ultimately, the goal of my research is to predict how anthropogenic drivers will alter disease risk in host communities. Central to achieving this goal is measuring linkages among community assembly and disassembly, interactions among parasites, and feedbacks that result from subsequent parasite epidemics. I do this by applying these ecological ideas to large datasets aggregated from multiple scientific studies.
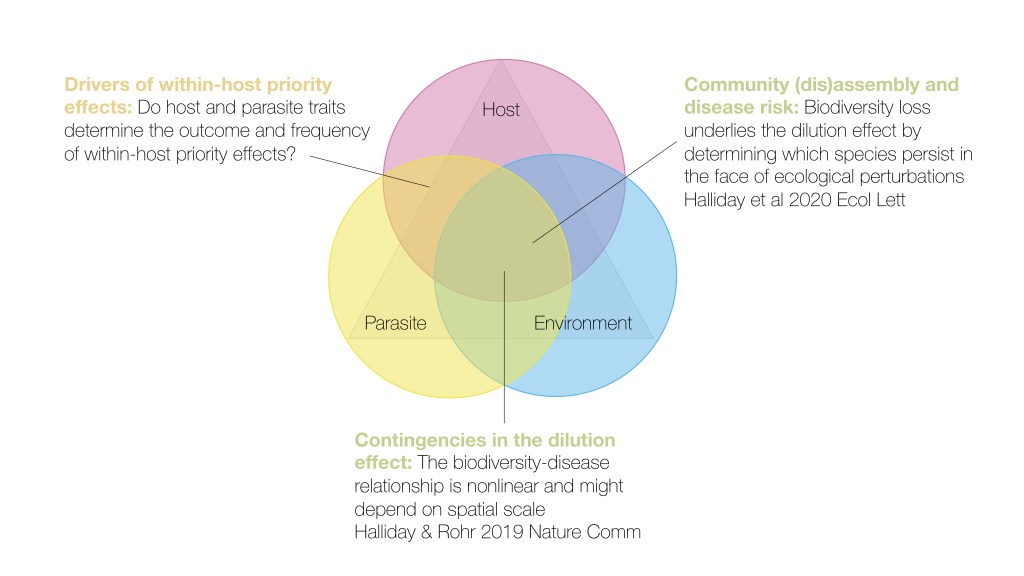
An important paper that was published in 2006 suggested that biodiversity may buffer host communities from infectious diseases under certain conditions. Yet, whether there is a general relationship between host diversity and disease risk has remained controversial. I use a meta-analytical approach to test for generality in the dilution effect of biodiversity. My past work suggests that biodiversity-disease relationships are commonly non-linear, might be scale dependent, and largely depend on whether biodiversity gradients are associated with a recent loss of species from local ecosystems.
I am also constructing a global database of coinfection studies across diverse host and parasite taxa that I will use to identify the characteristics of hosts and parasites that generate within-host priority effects. Please contact me if you would like to contribute to this database.
